06.02.2020
Heading in a new direction – Novel CARs specific for CD25 to treat chronic inflammatory processes
Life Sciences, Immunologie
- Chimeric antigen receptors to target chronic inflammation
- Therapeutic effect independent of tissue type and source
- Applicable to any inflammatory process
Ihr Kontakt
Dr. M. Charlotte Hemmer
- E-Mail:
- chemmer@baypat.de
- Telefon:
- +49 (0) 89 5480177 - 29
- Referenznummer:
- B79055
Factsheet
Download Tech Offer (PDF)Herausforderung
The last years have revolutionized cancer treatment with novel approaches using the patients’ own immune cells. CAR T cell therapy is furthest in clinical development and showed remarkable success in treating leukemia and advanced lymphomas1. Chimeric antigen receptors (CARs) are recombinant and modular trans-membrane receptors directed against different antigens and expressed on the surface of T cells via gene transfer. The genetically modified T cells are expanded ex vivo to then be re-infused back to the patient to act as a “living drug”. By binding to their targeted antigen, CAR T cells become cytotoxic and destroy the bound tumor cells. Knowledge on CAR T cells is now implemented into areas beyond cancer. In this context, chronic inflammation has come into focus. Common treatment options comprise the less effective nonsteroidal anti-inflammatory drugs and immunosuppressive steroids that act systemically and are associated with severe side effects. Specifically in autoimmune settings (e.g. in multiple sclerosis and rheumatoid arthritis) with the body’s immune system attacking its own tissues, the physiological function of regulatory T cells (Treg cells) to inhibit persistent inflammatory reactions, is not effective. There is a great need for more targeted therapeutics.
Innovation
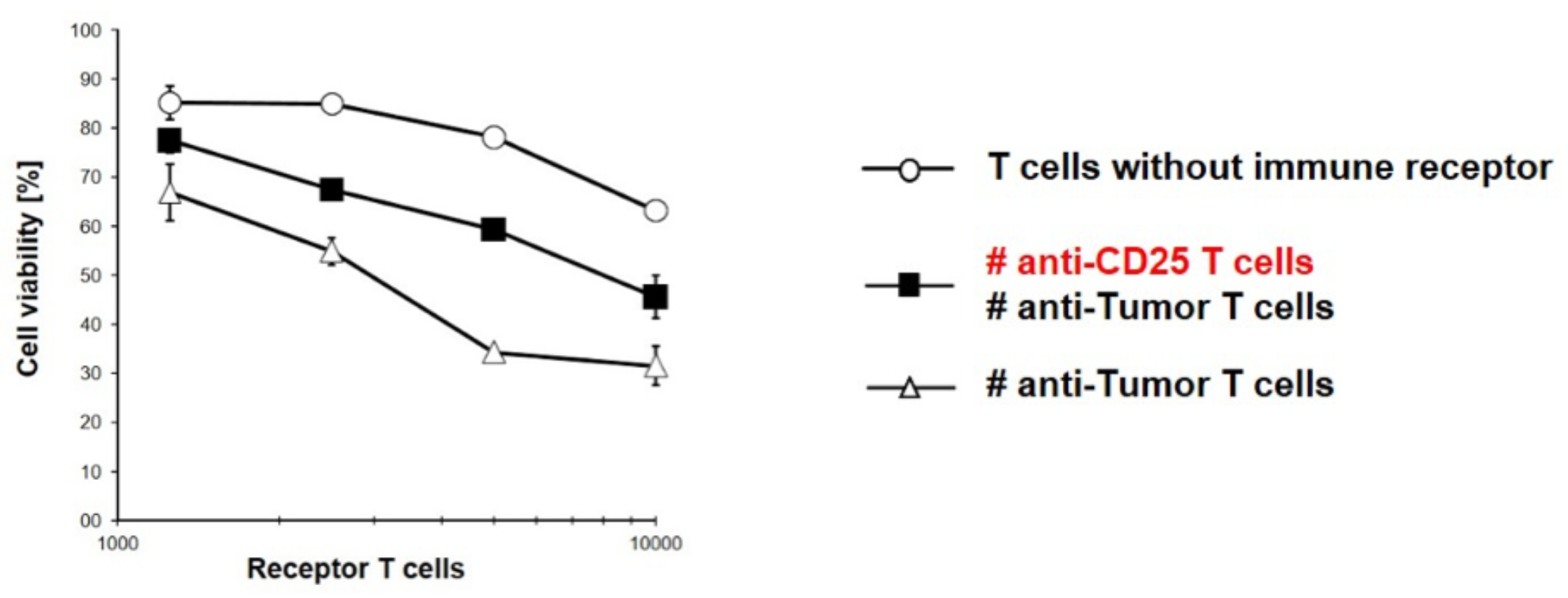
Our innovation applies the CAR T-cell technology in the context of chronic inflammatory processes. Here, CARs with specificity for CD25 (IL-2 receptor) were generated and, via gene transfer, expressed on cytotoxic T cells and suppressive Treg cells. Upon contact with activated immune cells, anti-CD25 T cells are able to diminish pro-inflammatory immune reactions by limiting tumor cell lysis and pro-inflammatory IL2-secretion independent of the tissue type. This approach can be used to control the persistent and de-regulated inflammatory response in autoimmune settings.
Kommerzielle Möglichkeiten
- High CD25-specificity for targeted suppression
- Anti-inflammatory effect is independent of tissue type
- Opportunities for partners include e.g. collaboration for in vivo studies
Entwicklungsstatus
Proof-of-concept in vitro.